integrated System for Trial Allocation and Randomisation (iSTAR) is a mobile-friendly web-based platform, used to streamline the processes of double-blind randomised controlled trials (RCT). iSTAR is designed to be flexible so that it can be customised as per specification. Trials that have used iSTAR: STRIDER Canada, OPSiP.
iSTAR offers the following features:
Screening & Randomisation
- Customised screening questions
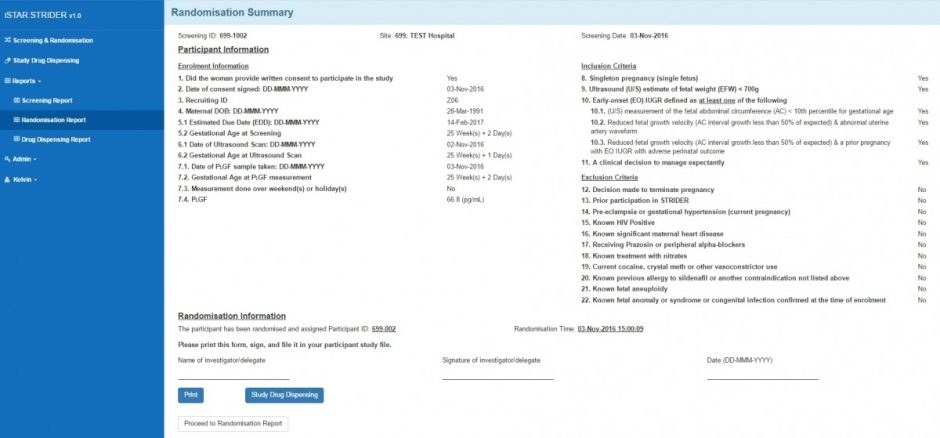
- Randomisation (random blocks and stratification)
- Participant Discontinuation – Stopping drug dispensing for participant
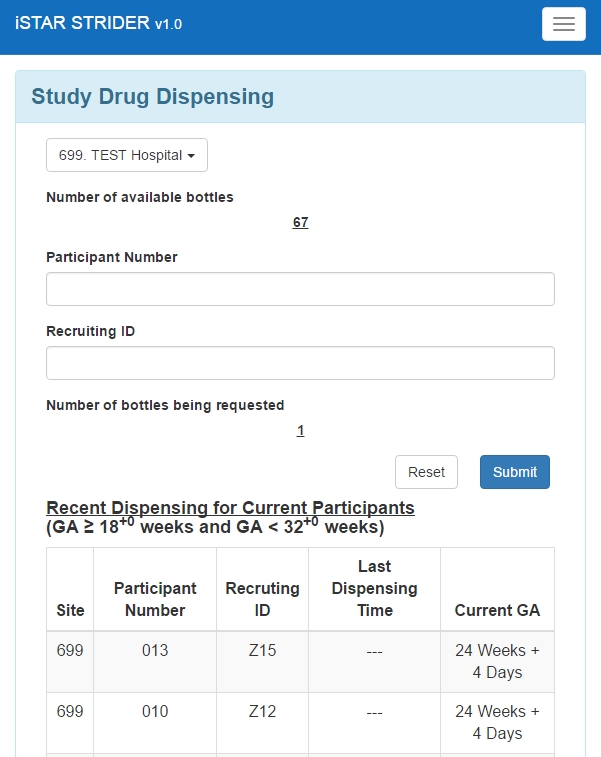
Drug Dispensing
- Multiple dispensing
- Information about bottles inventory per site
- Bottle/kit/lot number customisation
Bottle Management
- Setting bottle expiry date
- Inactivating bottles due to damage or loss, etc.
- Transferring bottles between sites
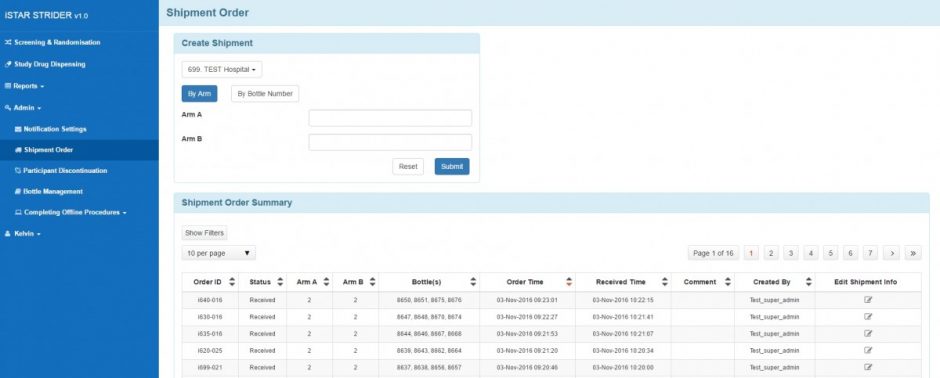
Bottle Shipment
- Creating, editing, and voiding shipment orders
- Shipment order summary report
Reporting Tool
- Screening, Randomisation, and Drug Dispensing reports
- Record filtering and sorting
- Report exporting in Excel
For Offline Procedures
- Manual entry of offline randomisation
- Contingency bottles dispensing
Email Notification
- Confirmation of randomisation and drug dispensing
- Notification of low inventory of study drug
Input Validation
- Real time validation
- Build-in algorithms for data validation
- Build-in algorithms for check of invalid data
User Access
- User groups assignment
- Separation of roles
- Access authentication and authorization
Configuration
- Email notification list
- Settings for randomisation/dispensing
- Settings for clinical trial/study
Error Handling
- Log of errors
- Log of security threats
- Log of locations and timestamps
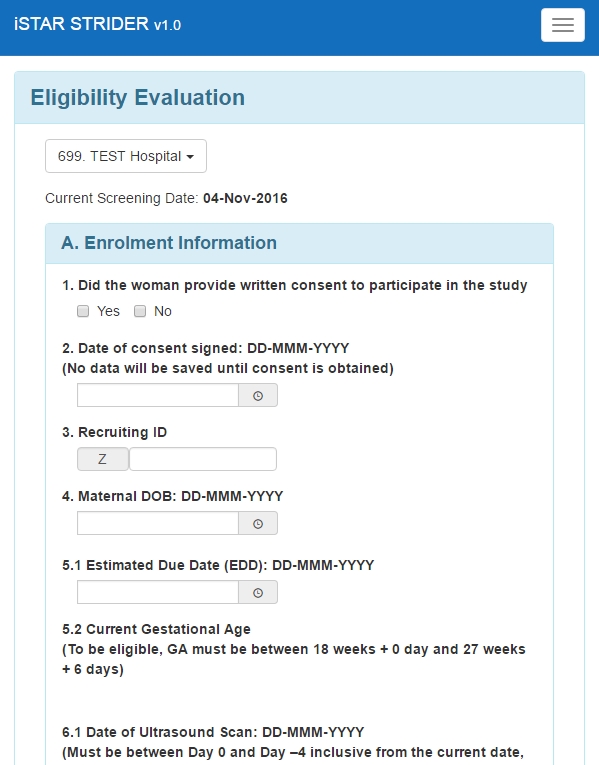
Mobile View – Eligibility Evaluation
Mobile View – Drug Dispensing
Randomisation Summary
Shipment Order